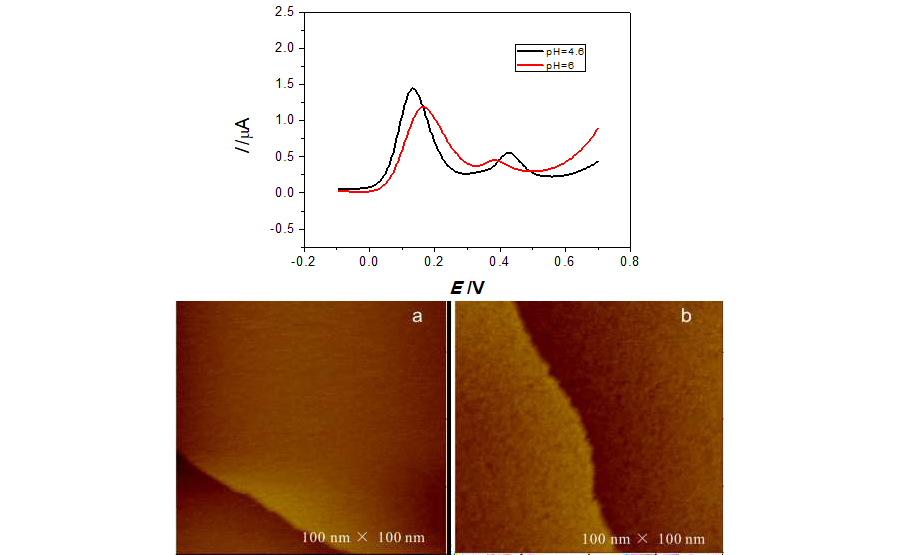
In this work, we have investigated the self-assembled monolayer (SAM) of L-cysteine on Au(111) and its application in electrochemical oxidation of uric acid by scanning tunneling microscopy (STM), cyclic voltammetry (CV) and differential pulse voltammetry (DPV). Monolayer of L-cysteine on Au(111) was observed, which shows that single monolayer of L-cysteine was formed on Au(111). Oxidation peak at 0.47 V was found for uric acid in 0.1 M HAc-NaAc buffer solution containing 2×10-5 M uric acid with pH=4.6 by CV, while two peaks can be observed for the solution with additional molecule of ascorbic acid. The influence of pH on electrochemical behavior of uric acid and ascorbic acid was also performed, which found that the peak distance and current is larger for pH=4.6 than that for pH=6. Those show that the solution with pH=4.6 may be good for the simultaneous detection of uric acid and ascorbic acid.